
When xsq inspection is in effect, tightened inspection shall be instituted when 2 out of 5 or fewer consecutive lots or batches have been non-acceptable on original inspection i. Under reduced inspection, the sampling procedure may terminate without making a decision. 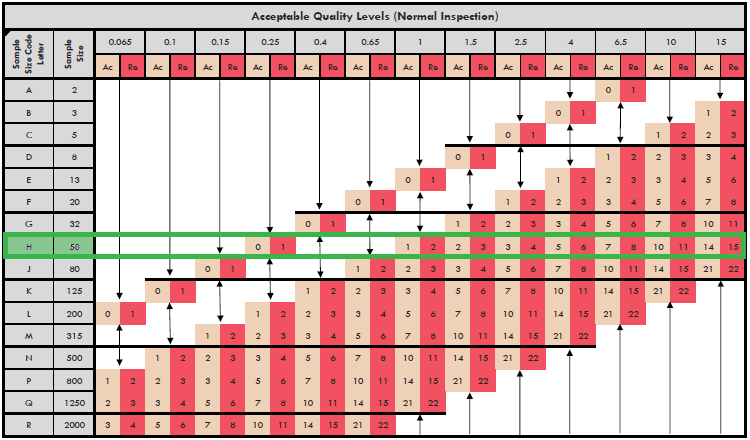 
If they want to know if individual units within the lot are acceptable - based on all criteria that is considered acceptable, then the tally of all defects found is correct. 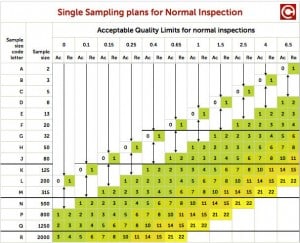
Sampling Procedures and Tables for Inspection by Attributes is one of the absi frequently used plans by many pharmaceutical companies as well as other industries. Attributes The FDA recognizes ANSI/ASQ Z as a General consensus standard. This e-standard is a very minor revision of ANSI/ASQ Z (R), also referred to as ANSI/ASQ Z ANSI/ASQ Z Sampling Procedures and Tables for Inspection By. ANSIASQZSampling Procedures and Tables for Inspection by Attributes- ANSI/ASQ Z Sampling Procedures and Tables for Inspection by.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
